Introduction
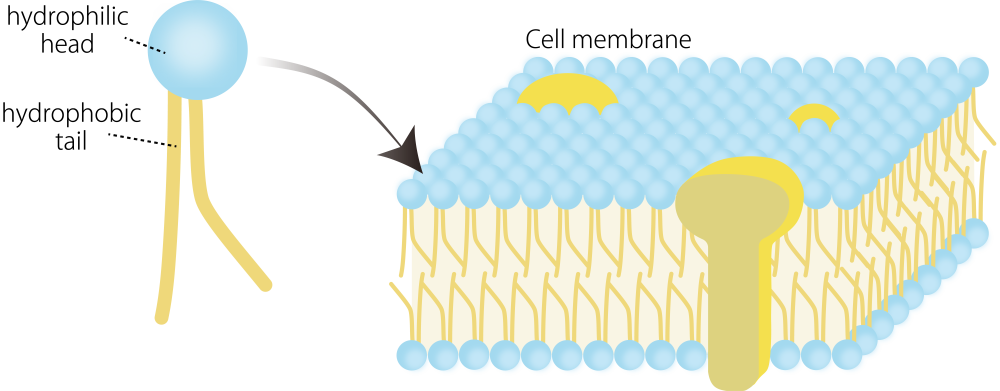
Phospholipids are the primary lipids that make up biological membranes and play essential roles in the construction and function of cellular and organelle membranes. Each phospholipid consists of a hydrophilic head and hydrophobic tails, and variations in their chemical structures result in a wide diversity of phospholipid species.
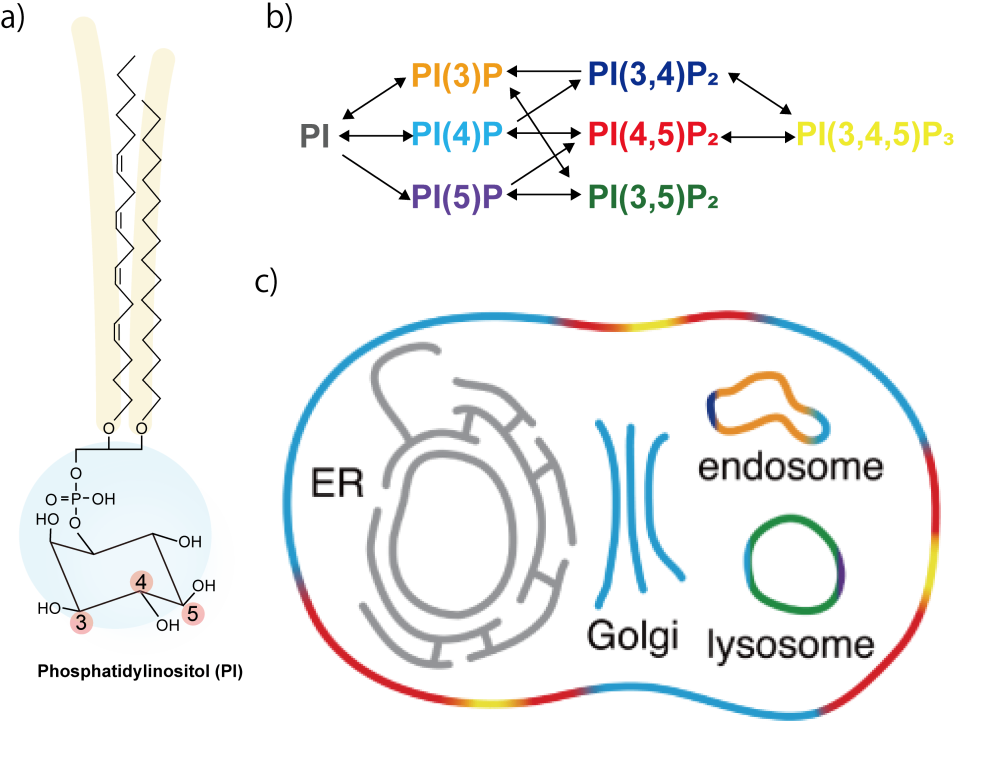
Among these, phosphoinositides are a representative class of phospholipids characterized by an inositol ring as their hydrophilic head group. There are eight major phosphoinositide species, defined by phosphorylation at the 3-, 4-, and 5-positions of the inositol ring (Figure 2a, b). These phosphoinositides are distributed throughout different intracellular compartments through precise spatial and temporal regulation by metabolic enzymes and transport proteins (Figure 2c). This asymmetric distribution imparts unique characteristics to each membrane, enabling the regulation of diverse cellular events such as signal transduction, membrane dynamics, cytoskeletal organization, and lipid transport.
Research Overview
Despite the tremendous diversity of lipids within biological membranes, the fundamental principles governing their spatial organization and contribution to membrane function—the “code of biological membranes”—remain largely unexplored. Our laboratory addresses this challenge by focusing on lipid metabolism and lipid transport.
1. Mechanisms of Spatiotemporal Regulation of Membrane Lipids
Lipid Metabolism
Enzymes responsible for lipid synthesis and degradation are tightly controlled in both their activity and localization, ensuring proper composition and distribution of lipids in the cell. However, the underlying mechanisms remain poorly understood. We focus on phosphoinositides, a functionally important class of lipids, to uncover how their metabolism is regulated at specific subcellular locations.
Lipid Transport
Membrane contact sites (MCSs), where cellular membranes are closely apposed, have emerged as critical hubs for lipid transport. Recent studies have shown that multiple families of lipid transport proteins mediate inter-organelle lipid exchange via these contact sites. Furthermore, our studies suggest that lipid transport across MCSs plays a pivotal role in the spatiotemporal regulation of lipid metabolism. We aim to uncover fundamental mechanisms governing membrane organization by dissecting lipid transport proteins, their cargo selectivity, and the regulatory basis of inter-organelle lipid transport.
2. Disease Mechanisms and Therapeutic Potential of Membrane Lipid Dysregulation
Lipid transport systems at membrane contact sites are increasingly recognized as key regulators of physiological processes including signaling, neuronal development, and pathogen life cycles. Dysfunction of these systems is associated with a range of diseases and infections. We aim to elucidate these mechanisms at the molecular, cellular, and organismal levels to understand how inter-organelle lipid transport contributes to physiology and disease, with the ultimate goal of enabling therapeutic development.
3. Decoding Lipid Dynamics in Living Cells
Unlike proteins, lipids are not directly encoded by genes and are inherently difficult to manipulate, which has hindered methodological advances in this field. In particular, imaging tools for visualizing and quantifying lipid localization and dynamics in living cells are crucial for understanding lipid function but remain underdeveloped. We develop imaging and analytical tools to visualize and quantify lipid dynamics in living cells, enabling real-time, high-resolution analysis of lipid behavior. These tools will also facilitate the identification and characterization of lipid dysregulation in disease contexts.

